About HPTN 083
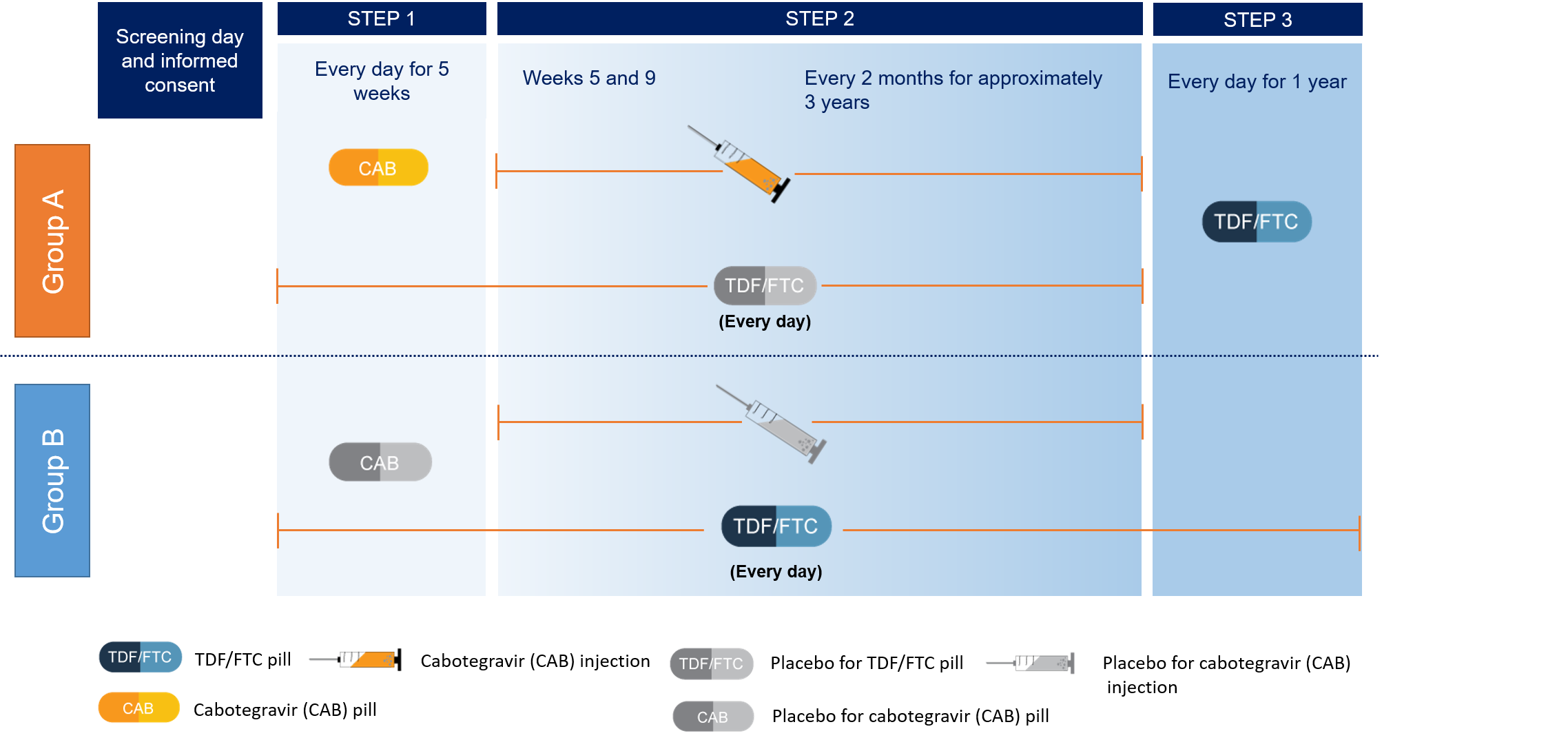
 HPTN 083 is the first large-scale clinical trial of a long-acting injectable drug with the potential to be used for HIV prevention. The study is examining whether a long-acting form of the anti-HIV drug cabotegravir injected once every 8 weeks can safely protect people from getting HIV infection at least as well as another anti-HIV medication combination taken as an oral tablet daily. The oral tablet, called Truvada, consists of the two anti-HIV drugs – emtricitabine and tenofovir disoproxil fumarate (TDF/FTC).
HPTN 083 is the first large-scale clinical trial of a long-acting injectable drug with the potential to be used for HIV prevention. The study is examining whether a long-acting form of the anti-HIV drug cabotegravir injected once every 8 weeks can safely protect people from getting HIV infection at least as well as another anti-HIV medication combination taken as an oral tablet daily. The oral tablet, called Truvada, consists of the two anti-HIV drugs – emtricitabine and tenofovir disoproxil fumarate (TDF/FTC).
Study Schema
Sponsored By:
Division of AIDS, US National Institute of Allergy and Infectious Diseases

